 It is then integrated with the grid by extrusion to form a plate. The oxide is mixed with water, sulphuric acid and a mixer, and then mixed to form a paste. This process would also increase the percentage of lead oxide in the material. This is produced from PbO by roasting in a flow of air. Red lead (Pb 3O 4) can also be added to the PbO formed by these methods, as it is more conductive. Ball milling: Pieces of lead are put into a rotary mechanical mill, forming fine lead flakes, which are then oxidised in air and removed.Each droplet reacts with the air to form an oxide layer, giving 70 – 85% lead oxide. Barton pot: A fine stream of molten lead is inserted into a heated vessel.The lead can be oxidised by two processes: The Barton pot and the ball mill. Grids can also be formed by mechanical working, either by cutting deep grooves into a sheet of steel, or by rolling up crimped strips and inserting them into holes in a cast plate, see Metal Forming TLP. The molds are closed and filled with sufficient molten lead to fill the mold, leaving some excess to form a sprue, which is then removed by cutting or stamping. Permanent steel molds are made from blocks by machining. “Book mold” casting is the most common method of production for the grid. The design is a simple grid framework with a “tab” or “lug” for connection to the terminal post. The function of the grid is to hold the active material and to conduct electricity between the active material and the battery terminals. A typical alloy would be 0.03 – 0.10% calcium and 0.5 – 1.0% tin (to enhance mechanical and corrosion properties). This is often used for telephone applications, and for no maintenance automotive batteries, since a more stable battery is required. (2) Alkaline earth metals such as calcium can be used to stiffen the lead. These act as grain refiners, decreasing the grain size of the lead and thereby increasing its hardness and strength. (1) Using below 4% the battery water consumption is reduced, however it is then necessary to add small amounts of other elements such as sulphur, copper, arsenic and selenium. There are two possible solutions to this problem: This means that the water consumption in the cell increases and frequent maintenance is necessary. However, during the operation of the battery the antinomy dissolves and migrates to the anode where it alters the cell voltage. Pure lead is too soft to use as a grid material so in general the lead is hardened by the addition of 4 – 6% antimony. The 6 cells add to give the 12V necessary to start a car.\( \newcommand\) The reverse reaction happens when the battery is being recharged. Since the electrons can't travel directly through the solution to get from where they're released to where they're used, the reactions can only continue if they travel through the external electrical circuit. Notice that one reaction releases electrons and the other uses them up. PbO 2 4H SO 4 - 2e -> PbSO 4 2H 2O and When the battery is being used, the 2 connections react to form lead sulfate (PbSO 4) by reacting with the sulfuric acid. In a car battery (sometimes called a lead-acid battery) the cathode is lead dioxide (PbO 2), the anode is a sponge of lead (Pb), and the solution is sulfuric acid (H 2SO 4). When a battery dies, it is because one or more of the chemical reactants is more or less used up. When run in reverse (with certain batteries), electrons are forced in the other direction in the reverse reactions. The electrons are incorporated in the products of this reaction. These reenter the battery through the cathode where another chemical reaction is happening between the cathode and solution. During normal operation, a chemical reaction occurs between the solution and the anode which releases electrons that flow through the circuit. There is also a solution that the cathode and anode sit in. There are 2 connectors that go out of the battery. Inside the battery are 3 important things. 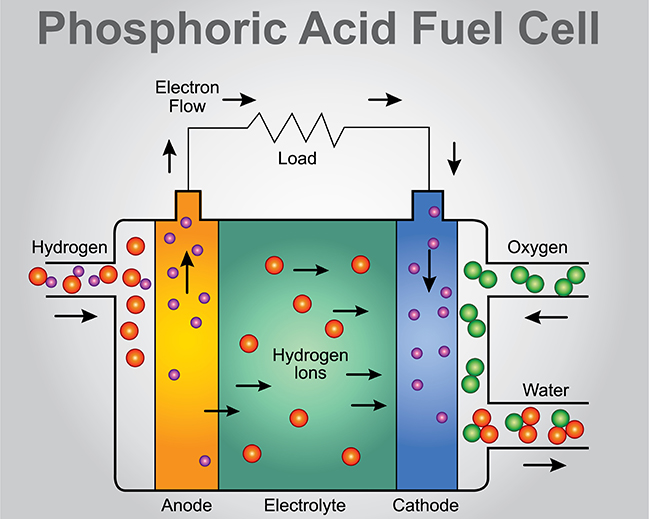
The general way that a battery works is that when an electronic circuit is connected to the battery, electrons are allowed to flow. This causes the voltages of each battery to add. A car battery is actually 6 smaller batteries that are lined up in series. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
